Preclinical Contract Research Organizations serve as essential partners in pharmaceutical development, validating drug safety and efficacy before human trials. The global preclinical CRO market reached $6.76 billion in 2025, projected to grow to $13.14 billion by 2034, reflecting increased industry reliance on specialized outsourcing.
These organizations conduct systematic evaluations of pharmacodynamics (drug effects on the body) and pharmacokinetics (how the body processes drugs). Core services include toxicology assessments, bioanalytical studies, and ADME profiling (Absorption, Distribution, Metabolism, Excretion) under FDA Good Laboratory Practices and ICH guidelines. By providing access to advanced infrastructure and scientific expertise, preclinical CROs enable pharmaceutical companies to reduce development risks and accelerate time-to-market while avoiding the costs of maintaining specialized facilities in-house.
What is a Preclinical CRO?
A preclinical CRO is a specialized contract research organization focused on conducting the essential studies required before pharmaceutical compounds and medical devices can proceed to human clinical trials. These organizations operate at the intersection of regulatory science and translational research, providing comprehensive services that span from early compound screening through IND-enabling studies required for regulatory submissions.
Preclinical CROs conduct both in vitro (test tube-based) and in vivo (living organism) studies to generate critical safety data. According to regulatory standards, preclinical testing must encompass multiple species, typically including rodent and non-rodent models, to establish no-observed-adverse-effect levels (NOAELs) that inform initial clinical dosing strategies. The strategic value of preclinical CROs extends beyond laboratory capabilities, as they serve as regulatory partners guiding pharmaceutical companies through complex FDA and EMA compliance requirements that vary across global markets.
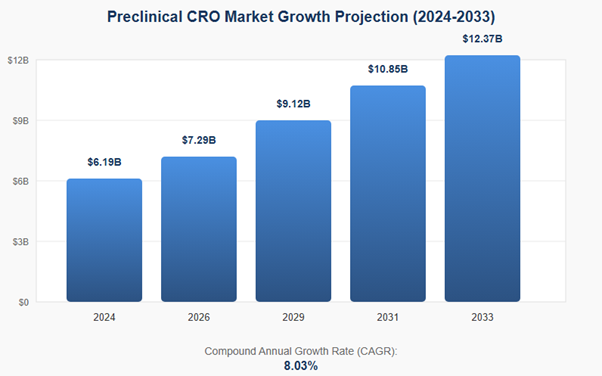
This represents consistent annual growth of 8.03%, indicating the market will nearly double from $6.19B to $12.37B over nine years. This strong growth rate reflects increasing pharmaceutical outsourcing demand and the rising complexity of drug development requiring specialized preclinical expertise.
Why Are Preclinical CRO Services Essential in Drug Development?
Pharmaceutical companies face development costs exceeding $2.6 billion per drug and timelines of 10-15 years, with only one in 5,000 compounds reaching approval. Preclinical CROs address these challenges by providing cost-effective outsourcing solutions that maintain rigorous scientific standards while reducing capital expenditures.
The critical need for CROs stems from high failure rates: 40-50% of drugs fail due to insufficient efficacy, 30% from toxicity issues, and 10-15% from poor pharmacokinetic properties. CROs mitigate these risks through specialized expertise in target validation, lead optimization, and safety profiling. Toxicology testing alone commands 51.6% of the preclinical CRO market share, reflecting its pivotal role in ensuring drug safety before human exposure.
Biopharmaceutical companies represent 81-82% of preclinical CRO service consumers, with small and medium-sized enterprises particularly dependent on outsourcing to advance their development pipelines.
What Types of Studies Do Preclinical CROs Conduct?
Preclinical CROs conduct six interconnected categories of studies that form the foundation of regulatory submissions. Toxicology testing evaluates potential adverse effects through acute, sub-chronic, chronic, reproductive, developmental, and genotoxicity assessments. Safety pharmacology studies examine effects on vital physiological systems including cardiovascular, respiratory, and central nervous systems to identify off-target effects that could compromise patient safety.
Bioanalysis and DMPK (Drug Metabolism and Pharmacokinetics) studies characterize how compounds are absorbed, distributed, metabolized, and excreted, establishing critical parameters for clinical dosing regimens. This segment is experiencing the fastest growth in the preclinical CRO market, with a projected CAGR of 8.5% through 2034, driven by increasing therapeutic complexity and regulatory emphasis on DMPK profiling as a predictor of clinical success.

The table shows key preclinical study types, their main objectives, and their 2024 market position. Toxicology testing dominates with 51.6% share, bioanalysis & DMPK is the fastest‑growing segment (8.5% CAGR), while chemistry & formulation is a growing area and safety pharmacology is described as an essential component of preclinical research.
Chemistry, manufacturing, and controls (CMC) activities ensure drug substance quality and stability, while analytical method development validates that testing procedures meet regulatory standards. For medical devices, biocompatibility testing evaluates whether device components are sustainable in living models, a critical consideration for implantable technologies and drug-device combination products.
How Do Large Animal Models Enhance Preclinical Research Accuracy?
Large animal models represent a critical component of preclinical validation, addressing the translational gap between rodent studies and human physiology. Regulatory agencies including the FDA and European Medicines Agency (EMA) specifically recommend large animal models for evaluating advanced therapeutic medicinal products (ATMPs), recognizing that species such as pigs, sheep, and calves share anatomical and physiological characteristics more closely aligned with humans than traditional rodent models.
Pigs have gained prominence as translational models due to proteomic, genomic, and immunologic similarities to humans. Their cardiovascular system closely mimics human physiology, with comparable heart rates (90-107 bpm in pigs versus 70-100 bpm in humans), similar coronary vasculature architecture, and analogous lipid profiles (60% LDL in pigs versus 63% in humans). These parallels make porcine models invaluable for cardiovascular device testing, gastrointestinal research, and metabolic studies including diabetes treatment development.
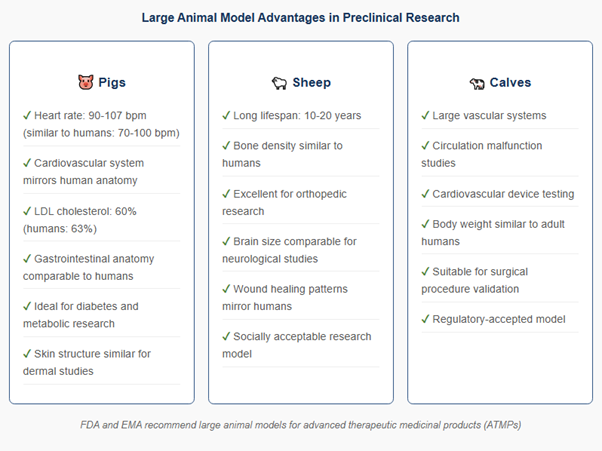
Why Large Animals? Unlike rodents, pigs, sheep, and calves share critical anatomical and physiological similarities with humans, including comparable organ sizes, cardiovascular systems, and metabolic processes. This makes them essential for predicting how drugs and devices will perform in human trials, significantly reducing the risk of late-stage failures that cost pharmaceutical companies billions.
Why Sheep and Large Animal Integration Matters?
Sheep excel in orthopedic research, wound healing studies, and respiratory evaluation. Their body size, organ proportions, and extended lifespans (10-20 years) enable long-term safety assessments that better predict human outcomes. Research in Frontiers in Bioengineering and Biotechnology demonstrates their value for cardiovascular disorders, bone regeneration, and reproductive medicine studies.
Regulatory authorities recommend a strategic step-wise approach: initial rodent screening followed by large animal validation that provides realistic late-stage development data. This methodology improves clinical translation success rates by identifying potential issues before expensive human trials commence, significantly enhancing the accuracy and reliability of preclinical research outcomes.
What Regulatory Standards Govern Preclinical CRO Operations?
Preclinical CROs operate under stringent regulatory frameworks to ensure study reliability and data integrity. The cornerstone is adherence to Good Laboratory Practices (GLP) as defined by the FDA (21 CFR Part 58) and international ICH guidelines. GLP standards mandate comprehensive documentation protocols, quality assurance systems, standardized operating procedures, and rigorous equipment calibration schedules.
For pharmaceutical compounds advancing toward clinical trials, preclinical CROs must generate data packages supporting Investigational New Drug (IND) applications. These submissions require comprehensive toxicology reports demonstrating acceptable safety margins, pharmacology data establishing mechanism of action, CMC documentation ensuring manufacturing quality, and clinical protocols detailing proposed human study designs. The IND review process by regulatory agencies directly determines whether compounds can proceed to clinical testing, making preclinical CRO data quality paramount.
Which Geographic Markets Lead Preclinical CRO Growth?
North America dominates the global preclinical CRO market with 47.5% market share in 2024, generating $2.26 billion in revenue. The United States accounts for 93.7% of this regional market, driven by concentrated pharmaceutical R&D investment, advanced research infrastructure, and substantial NIH funding that supports robust academic-industry collaborations.
The Asia-Pacific region represents the fastest-growing market, with projected CAGR of 10.9% through 2034. China leads this expansion by offering cost-effective services combined with expanding R&D infrastructure. India’s preclinical CRO market, valued at $183.3 million in 2023, is growing at 11.4% CAGR through 2030, positioning it as a strategic outsourcing destination for global pharmaceutical companies seeking to optimize development costs.
The preclinical CRO market is expected to grow 94% over the next decade, driven by pharmaceutical companies’ increasing reliance on specialized outsourcing partners to reduce development costs and accelerate time-to-market for new therapeutics.
European markets demonstrate steady growth supported by strong pharmaceutical sectors in the UK, Germany, Switzerland, and France. The European Federation of Pharmaceutical Industries reported R&D expenditure reaching $44 billion in 2022, reflecting sustained investment in drug development infrastructure and mature pharmaceutical industries.
What Emerging Technologies Are Transforming Preclinical Research?
Artificial intelligence and machine learning are revolutionizing preclinical drug discovery by accelerating target identification, compound screening, and toxicity prediction. In silico modeling enables researchers to simulate biological systems and predict drug behavior computationally before conducting expensive animal studies. AI-driven predictive modeling has demonstrated capability to reduce preclinical development timelines by 30-40% while improving success rates in identifying viable drug candidates. The National Center for Advancing Translational Sciences (NCATS) developed a comprehensive preclinical research toolbox in January 2024, incorporating AI-enabled resources that streamline drug discovery processes.
How Are Advanced Cell Culture Systems Changing Preclinical Testing?
Three-dimensional cell culture systems and organ-on-chip technologies provide physiologically relevant models that better replicate human tissue architecture and function compared to traditional 2D cell cultures. Organ-on-chip systems integrate engineered miniature tissues within microfluidic environments, enabling researchers to evaluate drug effects on multiple organ systems simultaneously. Patient-derived xenograft (PDX) and patient-derived organoid (PDO) models preserve tumor heterogeneity and genetic profiles from actual patients, enabling precision evaluation of therapeutic candidates. In September 2024, Hesperos’ Human-on-a-Chip technology supported Dianthus Therapeutics’ FDA Phase II trial submission, demonstrating regulatory acceptance of these innovative testing platforms.
How Do Preclinical CROs Support Specialized Therapeutic Areas?
Preclinical CROs deliver specialized expertise across distinct therapeutic domains requiring unique testing methodologies. Cardiovascular research utilizes large animal models particularly porcine and ovine to evaluate interventional devices, surgical techniques, and pharmaceutical interventions under conditions closely approximating human cardiac physiology. Organizations like Biotech Farm provide models examining blood circulation malfunctions, heart disease, and post-surgical complications that enable realistic device testing and procedure validation.
Diabetes and metabolic research represents another critical domain where large animal models evaluate glucose regulation mechanisms, insulin delivery systems, and metabolic interventions impossible to adequately study in rodent models. The complexity of diabetes pathophysiology, involving multiple organ systems and metabolic pathways, necessitates comprehensive preclinical assessment before clinical translation.
What Makes Orthopedic and Regenerative Research Unique?
Orthopedic and wound healing research relies on large animal models providing bone structures and tissue regeneration patterns comparable to humans. Sheep models prove particularly valuable for orthopedic device testing due to similar bone density and loading patterns. These models enable long-term evaluation of implant integration, bone regeneration, and healing processes under realistic biomechanical conditions, supporting development of next-generation orthopedic implants, bone graft materials, and tissue engineering products.
Specialized Preclinical CRO Capabilities
Organizations like Biotech Farm exemplify specialized preclinical CROs bridging laboratory research with clinical application through comprehensive large animal research services. Led by Adir Koreh with over seven years of research management experience, Biotech Farm operates state-of-the-art facilities accommodating pigs, sheep, and calves for complex disease modeling and medical device evaluation.
The facility specializes in diverse therapeutic applications across multiple physiological systems. Drug delivery research includes intravenous and topical application evaluation through pharmacokinetic studies establishing optimal dosing. Metabolic disease models support diabetes treatment development, while cardiovascular capabilities enable assessment of interventional devices and pharmaceutical interventions under controlled conditions predicting human outcomes.
Medical Device Testing and Integrated Solutions
Medical device testing at Biotech Farm encompasses post-surgical support devices for colon cancer patients, orthopedic implants, and bone regeneration technologies. Large animal models provide realistic anatomical environments for device placement and long-term integration assessment. Wound healing research leverages natural healing processes to evaluate advanced wound care products and regenerative medicine approaches.
The integration of experienced scientific teams with purpose-built facilities delivers comprehensive turnkey solutions. By maintaining in-house capabilities spanning surgical services, post-operative care, data analysis, and regulatory documentation, Biotech Farm accelerates pharmaceutical and medical device development timelines while ensuring regulatory compliance.
FAQ
What is the typical duration of preclinical studies?
Preclinical study duration varies significantly based on therapeutic complexity and regulatory requirements. Standard toxicology programs typically span 6-18 months, encompassing acute, sub-chronic, and chronic toxicity assessments. Comprehensive preclinical development programs including chemistry, manufacturing, and controls (CMC) work, DMPK studies, and GLP toxicology can extend 2-4 years before IND submission. Complex biologics or medical devices requiring long-term safety evaluation may necessitate even longer preclinical timelines.
How much do preclinical CRO services typically cost?
Preclinical development costs vary widely depending on study scope, duration, and complexity. Basic toxicology packages for small molecules may cost $500,000-$2 million, while comprehensive preclinical programs including multiple species, long-term studies, and specialized assessments can exceed $5-10 million. Large animal studies typically command premium pricing due to higher animal costs, specialized facilities, and longer study durations. The cost-benefit analysis favors outsourcing for most pharmaceutical companies, as establishing equivalent in-house capabilities would require capital investments exceeding $20-50 million plus ongoing operational costs.
What regulatory documents do preclinical CROs help prepare?
Preclinical CROs support preparation of comprehensive regulatory submissions required for IND applications. This includes Module 2 summaries providing written overviews of study findings, Module 4 nonclinical study reports containing detailed toxicology and pharmacology data, Module 3 CMC documentation establishing manufacturing quality and consistency, and clinical protocols detailing proposed human study designs. Many preclinical CROs provide regulatory affairs consulting to ensure documentation meets FDA, EMA, and other global regulatory authority requirements.
Why are both rodent and non-rodent studies required?
Regulatory agencies mandate testing in multiple species to increase confidence that safety findings translate to humans. Rodent studies (typically rats and mice) provide cost-effective screening for basic toxicity and efficacy. Non-rodent studies (commonly dogs, minipigs, or non-human primates) address species-specific differences in drug metabolism and provide data on larger physiological systems more comparable to humans. The FDA requires non-rodent toxicology data for most IND submissions, as some toxicities only manifest in non-rodent species. This multi-species approach reduces the risk of unforeseen adverse events in human trials.
How do preclinical CROs ensure data quality and reliability?
Preclinical CROs maintain data quality through rigorous GLP compliance, encompassing comprehensive standard operating procedures, regular equipment calibration and maintenance, independent quality assurance units that audit all studies, detailed documentation of all procedures and observations, and complete traceability of all study materials and data. GLP facilities undergo regular inspections by regulatory authorities to verify compliance. Many preclinical CROs also maintain accreditations from organizations such as AAALAC International, demonstrating commitment to animal welfare and scientific excellence.
What emerging therapeutic areas are driving preclinical CRO growth?
Gene and cell therapies represent the fastest-growing therapeutic area, requiring specialized preclinical testing including biodistribution studies, immunogenicity assessments, and long-term integration evaluation. mRNA therapeutics demand unique stability testing and delivery system validation. Personalized medicine approaches necessitate patient-derived models including PDX and PDO systems. Immuno-oncology combinations require complex immunological assessments in humanized mouse models. These advanced therapeutic modalities drive increasing demand for preclinical CROs with specialized capabilities beyond traditional small molecule testing.
Ready to Advance Your Drug or Device Development?
The preclinical phase establishes the foundation for clinical success through rigorous scientific evaluation and regulatory expertise. Specialized preclinical CROs provide strategic advantages through accelerated timelines, reduced costs, and improved success rates. Partnering with experienced research organizations ensures comprehensive evaluation under regulatory standards that global authorities demand. Facilities specializing in large animal research deliver translational insights that bridge laboratory discoveries and clinical application, reducing late-stage failure risks.
BIOTECH FARM supports your preclinical research needs with comprehensive large animal modelling services, expert scientific guidance, and regulatory-compliant study execution. Our state-of-the-art facilities and experienced team advance your therapeutic candidates toward clinical trials with confidence. Contact BIOTECH FARM today to discuss how our preclinical CRO services can accelerate your development program and position your innovations for regulatory success.






