ISO 10993 Preclinical Authority
Unveiling Biocompatibility Testing Animals: Navigating ISO 10993 for Implants and Preclinical Success
Authored by a team with over 30 years of combined expertise in large-animal surgical models and regulatory-ready biocompatibility evaluation — delivering scientifically defensible, ethically grounded preclinical results for medical device manufacturers worldwide.
FDA Compliant
3Rs Framework
Large-Animal Expertise
Expert Insight
Biocompatibility testing animals refers to the in vivo evaluation of medical devices and their materials in living biological systems, designed to confirm that a product does not provoke unacceptable adverse reactions when it interacts with tissues, blood, or organs. For implants and devices intended for prolonged or permanent contact, these studies form a decisive step in the safety dossier required by regulators worldwide — and a poorly designed study can cost months of rework.
Table of Contents ▼
2. When Are Animal Tests Truly Required Under ISO 10993?
3. Can Biocompatibility Be Achieved Without Animal Testing?
4. How to Select Endpoints for Preclinical Biocompatibility Testing
5. Understanding ISO 10993: The Rulebook for Biocompatibility
6. ISO 10993-2: The Ethics of Animal Welfare
7. ISO 10993-6: Implantation Study for Local Effects
8. ISO 10993-11: Systemic Toxicity Studies
9. In Vitro vs. In Vivo Biocompatibility
10. Common Animal Models for Implant Studies
11. Mapping Business Needs to Preclinical Capabilities
12. Timelines and Costs
13. Comparing Test Categories at a Glance
14. Frequently Asked Questions
15. Why Choose Biotech Farm?
Biocompatibility testing animals refers to the in vivo evaluation of medical devices and their materials in living biological systems, designed to confirm that a product does not provoke unacceptable adverse reactions when it interacts with tissues, blood, or organs. For implants and devices intended for prolonged or permanent contact, these studies form a decisive step in the safety dossier required by regulators worldwide.
An implant biocompatibility study becomes particularly relevant when chemical characterization, toxicological risk assessment, and in vitro screening cannot fully predict how a device will behave inside a whole organism. Systemic distribution of leachables, local tissue remodeling, capsule formation, or chronic inflammation are phenomena that often require a living model to be observed reliably. This is the essence of in vivo biocompatibility within a structured preclinical program.
What is In Vivo Biocompatibility Testing?
In vivo biocompatibility testing consists of animal studies designed to measure local tissue or systemic responses to a material, finished device, or extract, following a defined study design with predetermined endpoints aligned to ISO 10993 and regulatory expectations. Each protocol specifies species, route of exposure, observation periods, controls, and analytical readouts.
Typical examples within an implant biocompatibility study framework include local effects after implantation under ISO 10993-6, systemic toxicity according to ISO 10993-11, sensitization and irritation studies under ISO 10993-23, and pyrogenicity assessments. Each test answers a different biological question, and together they build a coherent picture of safety for the intended clinical use.
When Are Animal Tests Truly Required Under ISO 10993?
Animal testing is required when the biological risk assessment cannot be comprehensively addressed through existing data, chemical characterization, toxicological evaluation, and validated in vitro tests. Regulators advocate a hierarchical, stepwise approach: first leverage prior knowledge and material equivalence, then perform analytical and in vitro work, and only then progress to in vivo studies if relevant gaps remain.
The FDA guidance on ISO 10993-1 emphasizes that a Biological Evaluation Plan (BEP) must justify either the necessity of ISO 10993 animal testing or its absence. The final risk assessment must integrate the totality of available information, ensuring that biocompatibility preclinical decisions are evidence-based rather than reflexive.
Regulatory Principle
A Biological Evaluation Plan is not merely a checklist — it is a living scientific document that must proactively justify every test conducted and every test omitted, anchored in the device’s specific risk profile and intended clinical use.
Can Biocompatibility Be Achieved Without Animal Testing?
In selected scenarios, yes. When a strong scientific and regulatory justification covers all relevant biological endpoints, reliance on chemical characterization, structured risk assessment, peer-reviewed literature, material equivalence with previously approved devices, and validated in vitro assays may be sufficient. This pathway is increasingly recognized for low-risk devices and well-characterized materials.
The 3Rs principle – Replace, Reduce, Refine – is embedded in ISO 10993-2 and reinforced by institutional ethics frameworks such as the University of Zurich’s 3R framework. Regulatory bodies actively encourage minimizing animal use, and a thoughtful biocompatibility preclinical strategy can demonstrate that ISO 10993 animal testing is only used where scientifically indispensable.
???? Replace
Use non-animal alternatives — chemical characterization, in vitro assays, validated cell models — wherever scientifically valid and accepted by regulators.
???? Reduce
Minimize animal numbers through robust statistical planning, shared controls, and design efficiencies that answer multiple questions in a single well-powered study.
✅ Refine
Improve procedures to minimize pain, distress, and lasting harm — from optimized anesthesia and analgesia plans to early humane endpoints and enhanced post-procedural care.
How to Select Endpoints for Preclinical Biocompatibility Testing
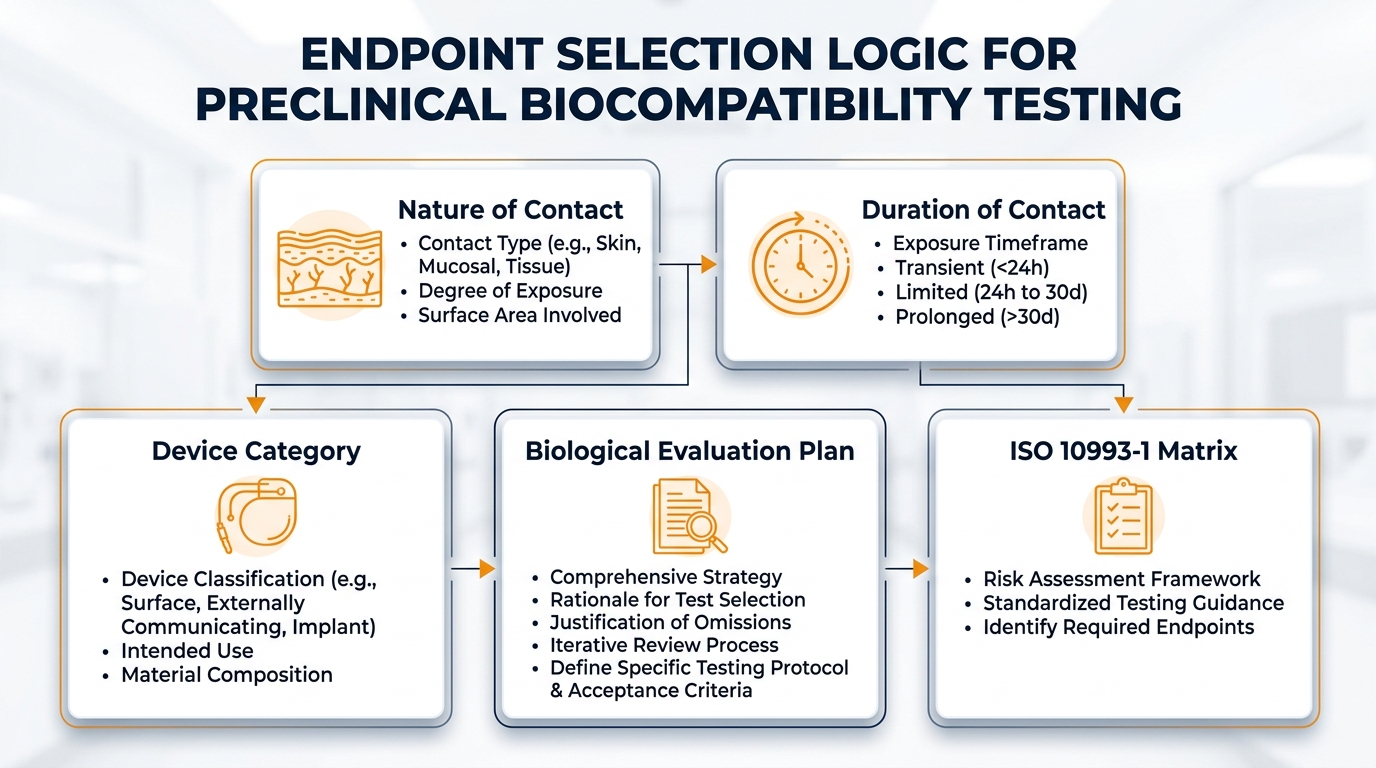
Endpoint selection is driven by several factors: the nature of tissue contact (skin, mucous membrane, breached surface, blood path indirect or direct, tissue/bone/dentin), the duration of contact (limited up to 24 hours, prolonged up to 30 days, permanent beyond 30 days), and the device category, including whether it is an implant. Each combination maps to a specific matrix of recommended biological evaluations within ISO 10993-1.
Accurate mapping is critical: it prevents costly over-testing, avoids unnecessary animal use, and reduces the risk of regulatory deficiency letters. A well-constructed plan integrates results from chemical characterization with targeted biocompatibility preclinical studies, ensuring every implant biocompatibility study and any ISO 10993 animal testing activity is scientifically justified. For more detailed information on creating a robust Biological Evaluation Plan, visit our Preclinical Testing Medical Devices Safety Evaluation page.
What Should a Robust Biological Evaluation Plan (BEP) Include?
A robust BEP includes a precise device description, full materials disclosure, manufacturing and sterilization processes, intended clinical use, contact category and duration, identification of toxicological hazards, gap analysis against ISO 10993-1, and clear justification for each chosen or omitted test. The FDA’s Basics of Biocompatibility resource center stresses that the BEP is a foundational element of the regulatory submission and must proactively address all foreseeable biological risks.
- Precise device description and intended clinical use
- Full materials disclosure including additives and processing aids
- Manufacturing flow, sterilization method and validation status
- Contact category and duration classification
- Gap analysis against ISO 10993-1 with justified test selection
- Prior biological, analytical, or clinical data supporting risk conclusions
Understanding ISO 10993: The Rulebook for Biocompatibility
ISO 10993 is a series of international standards governing the biological evaluation of medical devices. It provides a structured framework for selecting analytical, in vitro, and in vivo tests according to the device’s intended use, contact type, and contact duration, and it integrates these decisions into an overarching risk management process.
| Standard Part | Scope |
|---|---|
| ISO 10993-1 | Evaluation and testing within a risk management process |
| ISO 10993-2 | Animal welfare requirements |
| ISO 10993-6 | Tests for local effects after implantation |
| ISO 10993-10/23 | Irritation and sensitization |
| ISO 10993-11 | Systemic toxicity tests |
| ISO 10993-12 | Sample preparation and reference materials |
| ISO 10993-18 | Chemical characterization of medical device materials |
ISO 10993-2: The Ethics of Animal Welfare in Biocompatibility Studies
ISO 10993-2 codifies principles of animal welfare and demands ethical and scientific justification for any in vivo study. It operationalizes the 3Rs: Replace animal models with non-animal alternatives whenever scientifically valid; Reduce the number of animals to the minimum needed for statistical relevance; and Refine protocols to minimize pain, distress, and lasting harm.
These principles directly shape study design, influencing group sizes, time points, humane endpoints, anesthesia and analgesia plans, and post-procedural care. In Israel, animal studies are regulated by the Ministry of Health through the Council for Experiments on Animals, operating under the Animal Welfare Law (Experiments on Animals), 1994. This legal framework reinforces both the ethical foundation of biocompatibility preclinical work and the disciplined application of ISO 10993 animal testing.
“The 3Rs are not constraints on science — they are the foundation of rigorous, credible, and ethically defensible preclinical research.”
— ISO 10993-2 Core Principle
What is the ISO 10993-6 Implantation Study for Local Effects?
The ISO 10993-6 implantation study evaluates the local tissue response surrounding a material or device after surgical placement. Assessments include histopathology, acute and chronic inflammation, fibrosis and capsule formation, integration with surrounding tissue, neovascularization, and material degradation behavior over time.
The test sample is always compared against a control or reference material to distinguish surgical-site reactions from material-specific responses. Time points are selected based on the intended clinical use and the absorbable or non-absorbable nature of the material — ranging from short-term observations to long-term studies tracking degradation kinetics. The FDA underlines the importance of these evaluations, particularly for metal implants where inflammation, fibrosis, and necrosis must be carefully characterized as part of any rigorous in vivo biocompatibility assessment.
What Specific Histological Parameters Are Evaluated?
Evaluated parameters typically include polymorphonuclear cells, lymphocytes, plasma cells, macrophages, giant cells, necrosis, neovascularization, fibrosis, fatty infiltrate, and material remnants. Standardized scoring follows the guidance within the ISO 10993-6 standard, ensuring inter-study and inter-laboratory comparability of histopathological interpretation.
???? Acute Indicators
- Polymorphonuclear cells
- Necrosis
- Giant cells (early)
???? Chronic Indicators
- Lymphocytes, plasma cells
- Fibrosis, capsule formation
- Fatty infiltrate, macrophages
???? Integration Signs
- Neovascularization
- Tissue ingrowth
- Material remnant analysis
How Are Implantation Site and Animal Model Selected?
Selection is governed by clinical relevance: subcutaneous tissue, muscle, bone, or specific anatomical sites that mimic the intended use. Sample geometry, available implant size, ability to harvest tissue with adequate margins, and the need to compare multiple groups or time points all influence the decision. A well-chosen model strengthens the predictive value of any implant biocompatibility study and the overall in vivo biocompatibility conclusion.
Exploring ISO 10993-11: Systemic Toxicity Studies
ISO 10993-11 evaluates whether substances released from a device — leachables and extractables — can cause whole-body toxic effects after acute or prolonged exposure. Studies typically include extract preparation per ISO 10993-12, administration via clinically relevant routes, clinical observations, body weight and food consumption monitoring, hematology, clinical chemistry, and gross and microscopic pathology where appropriate.
This pillar of biocompatibility preclinical evaluation complements local-effects testing by addressing organs and systems distant from the implantation site. Properly designed ISO 10993 animal testing for systemic effects gives regulators confidence that no hidden systemic hazard accompanies the device’s intended use.
Acute vs. Subacute vs. Subchronic vs. Chronic: Understanding the Differences
| Study Type | Duration | Primary Focus |
|---|---|---|
| Acute | ≤24 hours | Single-exposure immediate effects |
| Subacute | Up to 28 days | Repeated-dose short-term toxicity |
| Subchronic | Up to 90 days | Extended exposure with clinical pathology |
| Chronic | >90 days | Long-term systemic effects, full histopathology |
The Difference Between In Vitro and In Vivo Biocompatibility (and When to Combine Them)
In vitro tests provide focused, rapid screening using cells or tissue models — cytotoxicity, hemolysis, genotoxicity in cell systems — delivering early signals on material safety. In vivo biocompatibility studies, by contrast, assess complex, integrated biological responses in a whole organism, capturing systemic effects, tissue remodeling, and time-dependent phenomena that cell cultures cannot reproduce.
A combined strategy minimizes risk and ensures animal studies are conducted only where in vitro data and analytical evidence leave genuine gaps. This integrated logic saves time and budget, prevents repeated rounds of testing, and aligns the biocompatibility preclinical program with the 3Rs.
Best Practice
Always begin with comprehensive chemical characterization and in vitro screening. Progress to in vivo studies only when analytical and cell-based data identify gaps that cannot be closed without a whole-organism model. This sequencing is both scientifically stronger and more cost-efficient.
Common Animal Models for Implant Biocompatibility Studies
Model selection depends on the implantation site, implant size, intended follow-up duration, and whether the target tissue is bone, muscle, vascular, or soft connective tissue. Larger animal models are often preferred when implant dimensions, anatomical relevance, or long-term observation align with human clinical conditions. Smaller models may be sufficient for screening or short-term studies.
The chosen species and site must support reproducible histological analysis and meaningful comparison across groups and time points. Robust biocompatibility testing animals programs select models that balance scientific validity, ethical considerations, and translational value, ensuring each implant biocompatibility study generates credible in vivo biocompatibility data.
Mapping Business Needs to Preclinical Capabilities

Manufacturers benefit when a preclinical partner aligns its operational structure with sponsor needs across planning, execution, and reporting. The table below illustrates how typical business needs map to practical capabilities at a structured large-animal facility.
| Business Need | How an Integrated Preclinical Facility Supports It |
|---|---|
| Tailored study design | Joint protocol development with senior scientific staff, matching ISO 10993 endpoints to the device’s intended use |
| Regulatory readiness | Documentation aligned with FDA expectations and ISO 10993-1 risk management framework |
| Ethical compliance | Adherence to the 3Rs and to the Israeli Animal Welfare Law (Experiments on Animals), 1994 |
| Surgical complexity | Equipped operating rooms with imaging, fluoroscopy, ultrasound, and laparoscopic capabilities |
| Long-term studies | Animal housing designed for prolonged welfare and consistent monitoring over weeks to months |
| Transparent collaboration | Interactive review meetings, clear documentation, and continuous sponsor access during the project |
How Long Does Preclinical Biocompatibility Testing Take?
Timelines vary substantially with the test matrix, device complexity, and regulatory pathway. Acute systemic and irritation tests may be completed in a few weeks, while subchronic and chronic implantation studies often require several months from protocol approval to final report. Add to this the lead times for ethics committee approval, animal acclimatization, histopathology turnaround, and regulatory peer review.
Realistic planning starts with a comprehensive gap analysis: extensive chemical characterization, custom surgical models, and long-duration follow-up all extend the schedule. A disciplined biocompatibility preclinical roadmap, agreed early with the testing facility, prevents bottlenecks and aligns deliverables with submission milestones.
The Cost of ISO 10993 Biocompatibility: What to Expect
Cost drivers include the number and type of tests, species and number of animals, study duration, surgical complexity, imaging requirements, histopathology depth, and any regulatory consulting needed to interpret results. Long-term implantation studies and multi-time-point designs naturally carry higher costs than acute extract-based tests.
A well-structured BEP is the most effective cost optimizer. Strategic use of existing data, material equivalence arguments, robust chemical characterization, and well-chosen in vitro screens can reduce the volume of ISO 10993 animal testing required, focusing investment where it delivers the highest scientific and regulatory value.
Cost Risk Alert
Poorly scoped test matrices are the leading cause of unexpected cost overruns in biocompatibility programs. A proactive BEP reviewed by an experienced preclinical partner before study initiation consistently delivers better budget predictability and avoids costly repeat studies.
Comparing Test Categories at a Glance

The following comparison summarizes the scope and typical role of each major ISO 10993 test category in an integrated evaluation program.
| Test Category | Primary Question | Typical Approach |
|---|---|---|
| Chemical characterization (ISO 10993-18) | What can leach from the device? | Analytical chemistry on extracts |
| Cytotoxicity (ISO 10993-5) | Are extracts toxic to cells? | In vitro cell-based assay |
| Sensitization & Irritation (ISO 10993-23) | Does the device provoke immune or local reactions? | In vivo, with extract or direct contact |
| Implantation (ISO 10993-6) | How does tissue respond locally over time? | In vivo surgical implantation with histology |
| Systemic toxicity (ISO 10993-11) | Are there whole-body effects? | In vivo extract administration with clinical pathology |
Our Methodology: Preparing for Your Biocompatibility Testing
For an accurate quotation and an efficient timeline, manufacturers should provide a clear device description, complete materials list including additives and processing aids, manufacturing flow, sterilization method and validation status, intended clinical use, contact type and duration, product drawings or CAD files, and any prior biological, analytical, or clinical data.
The more transparent the input package, the more precisely a biocompatibility preclinical partner can tailor the BEP and design each implant biocompatibility study. Sponsors who supply early-phase chemical characterization and risk assessment outputs typically receive sharper test matrices and tighter cost estimates.
Ensuring Regulatory Compliance: Our Expertise in Biocompatibility
Compliance is not a single checkbox — it is a continuous alignment between study design, execution quality, and the expectations of bodies such as the FDA, notified bodies under the EU MDR, and Israeli regulators. A capable preclinical partner maintains current literacy in ISO 10993 revisions, FDA guidance updates, and evolving expectations around chemical characterization and toxicological risk assessment.
A risk-based approach drives every plan: identify hazards, evaluate exposure, prioritize the evidence that closes the highest-risk gaps, and avoid redundant ISO 10993 animal testing. The result is a biocompatibility preclinical program that is both scientifically defensible and operationally efficient.
Why Choose Biotech Farm for Your In Vivo Biocompatibility Studies?
Biotech Farm is a state-of-the-art large-animal preclinical facility led by a team with more than 30 years of experience in research and project management. Surgical suites are equipped with C-Arm fluoroscopy, high-definition ultrasound, echocardiography, surgical microscopes, and 4K laparoscopic systems — supporting complex implantation and follow-up procedures relevant to demanding in vivo biocompatibility studies.
Beyond infrastructure, sponsors benefit from a tailored scientific escort, transparent collaboration, well-documented procedures, and uncompromising attention to animal welfare under the 3Rs framework. The commitment is clear: deliver accurate, regulator-ready results that advance every implant biocompatibility study efficiently and ethically.
????
Advanced Surgical Suite
C-Arm fluoroscopy, HD ultrasound, echocardiography, surgical microscopes, and 4K laparoscopic systems for complex implantation procedures.
????
Scientific Escort
Tailored study design, transparent documentation, and interactive sponsor collaboration from BEP through final report delivery.
????
Animal Welfare First
Full 3Rs compliance, ISO 10993-2 adherence, and oversight under the Israeli Animal Welfare Law for every study we conduct.
Frequently Asked Questions About Biocompatibility with Animals
Are all medical devices required to undergo animal testing? ▼
What is the role of the 3Rs in animal biocompatibility testing? ▼
How does chemical characterization relate to in vivo animal tests? ▼
What is the typical duration for an implant biocompatibility study? ▼
Can an existing implant rely on a “shortcut” for biocompatibility? ▼
Does Israeli regulation accept ISO 10993 and FDA frameworks? ▼
Ready to Plan Your In Vivo Biocompatibility Program?
Are you preparing a regulatory submission and wondering which biocompatibility testing animals studies are truly necessary for your device? Our team is ready to review your BEP, map your endpoints to ISO 10993, and design an implant biocompatibility study tailored to your timeline and budget.






